Hot off the press! Our latest work on photochemical activation of light alkanes just appeared in Science (https://doi.org/10.1126/science.abb4688). We are stoked to present our group’s first Science paper to the community. In this blog article, we would like to discuss the human story behind this amazing project.
New project, new challenge
After our first project on the C(sp3)–H oxidation using decatungstate-photocatalysis, published almost two years ago in Angewandte Chemie (https://doi.org/10.1002/anie.201800818), we were looking for a new challenge where we could exploit the potential of this potent hydrogen atom transfer (HAT) catalyst. The NRG-TBADTeam (nickname of the subgroup dedicated to TBADT chemistry) focused its attention to a valuable -yet absent in organic synthesis- class of gasses which could benefit from continuous-flow microreactor technology: the volatile alkanes, such as methane, ethane, propane and butane. These compounds are mostly burned to release its energy for heating applications or propulsion, while in the chemical industry they are used as inexpensive starting materials for the synthesis of haloalkanes or polymer monomers (e.g. ethylene). Hence, we wondered if we could activate these inert and insoluble gasses with TBADT and immediately engage them in synthetically-useful transformations of interest to the organic chemistry community. Such a strategy would allow us to bypass the current toxic halogenation industry.
We commenced our investigations with propane and borrowed a bottle of high purity propane from our TU/e colleague Prof. F. Gallucci. To our delight, we could immediately spot some interesting reactivity. We were particularly delighted by the excellent selectivity provided by decatungstate for the most substituted carbon of propane (86:14 ratio for the instalment of a secondary propyl versus primary propyl moiety). At that point, master student Klaas van der Wal joined the group and together with him the real optimization commenced. Klaas did an amazing job, especially in the design of the flow setup, which was crucial to bring the gaseous reactants into contact with the soluble catalyst and substrates, and the initial reactivity screening. He received a great score for his MSc defense (Figure 1). However, we were not done yet!


Figure 1. TU/e M.Sc. student Klaas in action: left) at the Master thesis presentation and defence; right) graduation ceremony.
Shine brighter
Even if an extensive optimization was already conducted at that point, we managed to run our reactions only in stop-flow mode to reach full conversion. We realized that a more powerful light source was needed to reduce the reaction time. More photons were going to be crucial in order to move to a continuous operation of the flow reactor. For this reason, we decided to employ a Vapourtec setup, which was provided to us by Vapourtec’s CEO Duncan Guthrie and Dr. Manuel Nuño. Thanks to this photochemical flow system equipped with a powerful 365 nm LED set (60 W or 150 W, the latter high power 365 nm light source was even a prototype which proved crucial in our reaction discovery), we could easily move to a continuous-flow mode and complete our last screening.
At that point, Tim asked Yuchao Deng, who was a visiting Chinese PhD student in our lab, to join the TBADTeam. With her hard work and dedication, we were able to rapidly carry out the propane optimization and to identify the substrate scope. During the scope exploration, the input from Profs. Maurizio Fagnoni and Davide Ravelli was of great value to make rapid progress. They have been working with this catalyst for many years and TBADT has essentially no secrets for them anymore.
Next, we evaluated a variety of different alkane gases, starting with isobutane. Activation of isobutane led to a new methodology which allowed to introduce tert-butyl groups in a straightforward and selective fashion (96:4, tert butyl versus isobutyl).

Figure 2. Gabriele and Yuchao working with the Vapourtec setup.
Methane? Yes, we can!
We must acknowledge that at point we were quite satisfied with the results but we were still anxious as two formidable challenges laid ahead of us: the activation of ethane and methane. These alkanes possess the strongest C(sp3)–H bonds known in Nature and we were not sure whether decatungstate would be able to cleave those selectively… Methane for example has been a daunting challenge for many decades, occupying many researchers without much success so far. Cleaving methane’s C(sp3)–H bonds requires extremely high temperatures (> 500 C) and is only industrially done for a few processes.
For ethane, we needed to crank up the pressure a bit to get it into a liquid state. However, overall the activation went pretty smooth and only minor optimization was required to obtain a satisfactory scope.
Finally, we were ready to take on the challenge of methane. If this one would work, we knew it would be a big deal. And … we had probably a good shot to get it into a true top journal. In our first attempts, we could spot the right product only in traces. We realized that the major products obtained were propylated adducts or compounds derived from the activation of acetonitrile. The presence of propylated compounds puzzled us for a while. However, we realized that the gas line might still contain some propane from our previous experiments. Indeed, by applying vacuum on the gas lines, we could remove traces of condensed propane fairly quickly. With propylation being resolved, we saw that the HAT on acetonitrile was still prominent, and only a low yield was observed for the desired methylated product. We then realized that activation of acetonitrile was favoured over the activation of methane. Even if this result could be expected based on BDE, we originally thought that the polarity mismatch of the C-H bonds of acetonitrile would prevent this side reaction. This was indeed the case for butane, propane and ethane but not for the strongest C–H bonds present in methane. To circumvent this, we employed d3-acetonitrile. Once we did that, we finally obtained the desired products in synthetically useful yields. After isolating our first methylated molecule, Yuchao and I were so happy that I cannot even describe it in words. We repeated at least a thousand times “WE DID IT!” jumping all over the lab. Definitely, that was not a quiet day for our labmates.
Hard times, high hopes
When we were wrapping up our experiments, the pandemic crisis was advancing very fast. As an Italian, I was extremely aware of the potential threat of COVID-19. Yuchao was even more scared than I was, especially because her time in the NRG group was almost over and she would not be able to go back to China as her flights got systematically cancelled. Despite the COVID stress, we completed all the necessary experiments. Tim wrote the paper and submitted it to Science on February 26, a couple of weeks before the inevitable lockdown of our group (March 13, 2020). Then we waited, isolated at home.
On April 7, we got news! We received a request to submit a revision, so not rejected. What a relief! After reading the feedback from the referees, we were cautiously optimistic. Yet we were also scared! We felt we could smell our first paper in Science and we did not want to mess it up. After some Zoom calls with all co-authors, we submitted a carefully revised version and waited again, all of us still sheltered in place. On May 6, 2020, we finally got the acceptance letter (Figure 3, Top). As the email came in quite late in Europe, Tim was already sleeping. On May 7 at 6.20 AM, he send me via WhatsApp some very exciting messages (Figure 3, Bottom).
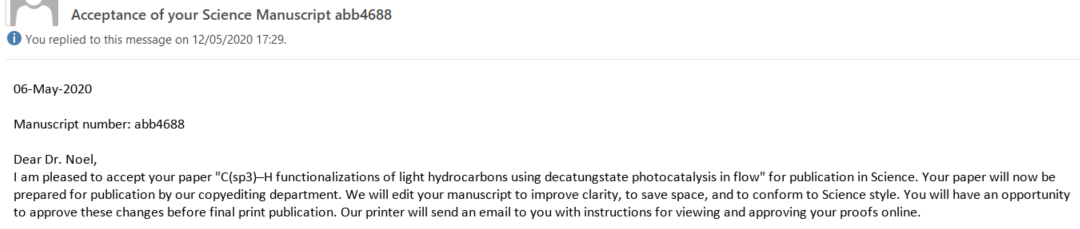
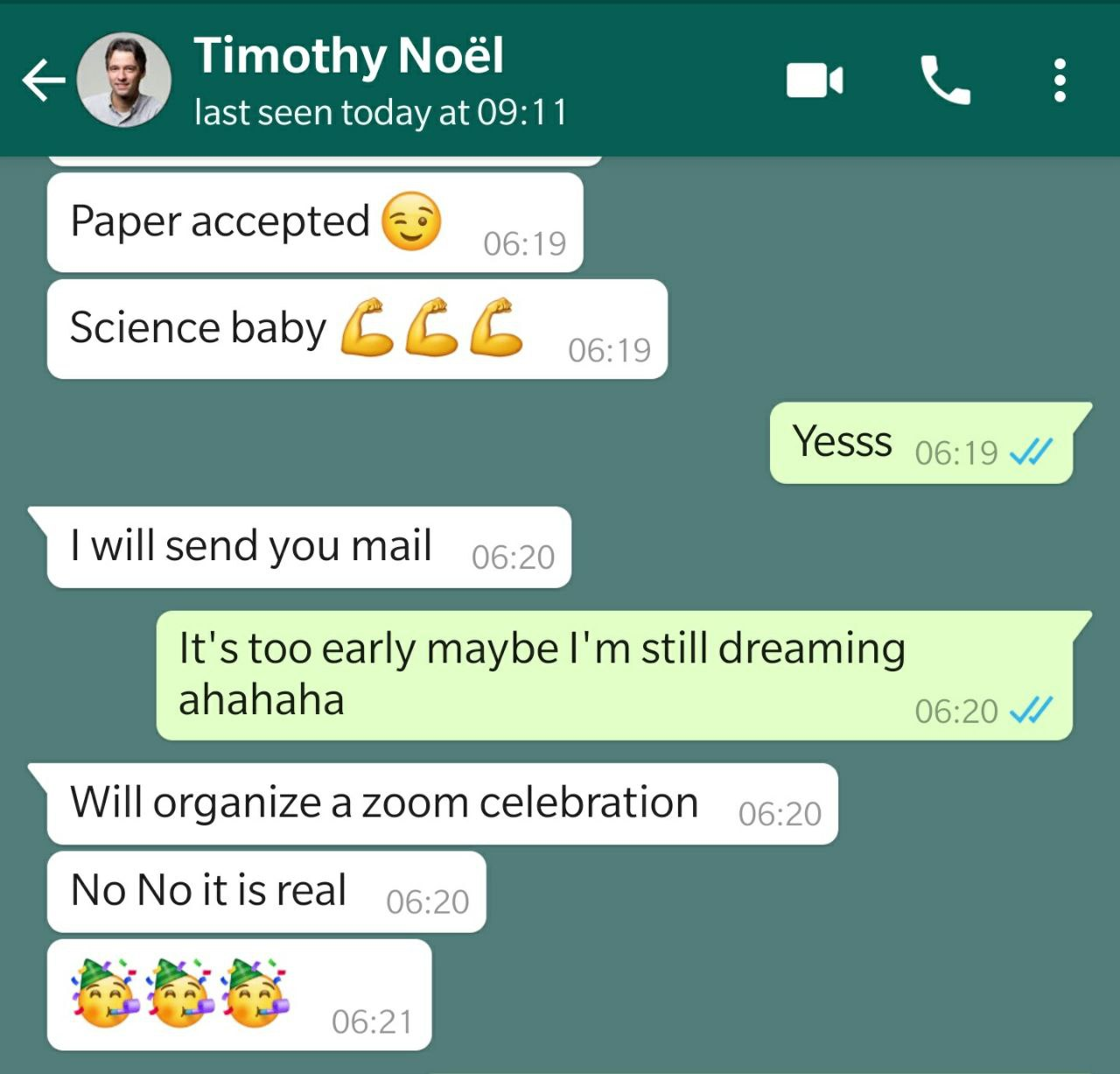
Figure 3. Top: Email with the long awaited final decision. Our first paper in Science is a fact. Bottom: WhatsApp conversation between Tim and myself (Gabriele Laudadio).
We are now July 2nd, almost two months later. The paper just got published in the latest issue of Science. But I still need to pinch myself regularly to know I am not dreaming. I will cherish these memories forever.
Ciao,
Gabriele
The paper discussed in this blog was published as “C(sp3)–H functionalizations of light hydrocarbons using decatungstate photocatalysis in flow”. by Gabriele Laudadio, Yuchao Deng, Klaas van der Wal, Davide Ravelli, Manuel Nuño, Maurizio Fagnoni, Duncan Guthrie, Yuhan Sun, and Timothy Noël, Science 2020, DOI: 10.1126/science.abb4688.
